WHY OXBRYTA?
TULLYHigh school senior, trumpet player, and actual Oxbryta patient
HOW OXBRYTA WAS STUDIED
Oxbryta Tablets and Oxbryta 300 mg Tablets for Oral Suspension were studied in 2 different age groups in 2 clinical trials: HOPE and HOPE-KIDS 1.
THE HOPE TRIAL STUDIED PATIENTS AGES 12 YEARS AND up
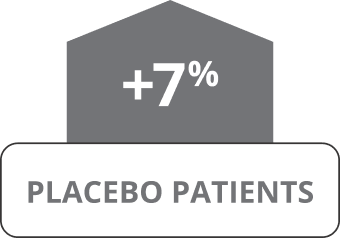
Oxbryta Tablets were studied in 90 patients who received Oxbryta (daily dose of 1,500 mg) and 92 patients who received a placebo (sugar pill) for 24 weeks.
The goals of this study
The main goal of the HOPE clinical trial was to determine if treatment with Oxbryta helped increase hemoglobin by more than 1 gram per deciliter (g/dL) through 24 weeks of treatment.
Another goal was to determine if Oxbryta also helped decrease hemolysis.
- More than half of patients (65%) were taking hydroxyurea along with Oxbryta and continued to do so during the entire trial
- 70% of Oxbryta patients continued through 72 weeks
OXBRYTA SIGNIFICANTLY INCREASED
HEMOGLOBIN LEVELS
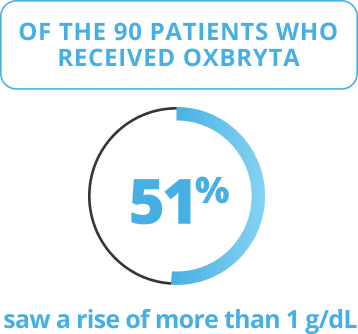
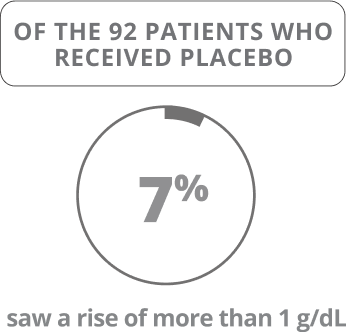
Results shown were at 24 weeks.
Oxbryta significantly DECREASED hemolysis
When fewer red blood cells break down, anemia improves. Some ways to know anemia has improved:
- Lower bilirubin Bilirubin [bil-uh-roo-bin] is a substance made in the body when red blood cells break down. Low bilirubin levels are good because that means fewer red blood cells are breaking down, which can lead to anemia. levels
- Lower reticulocyte A reticulocyte [ri-tik-u-lo-site] is a new or “baby” red blood cell that has not yet fully developed. When your body isn’t getting enough oxygen, the body works hard to add more red blood cells by creating reticulocytes. counts
On average, bilirubin levels decreased
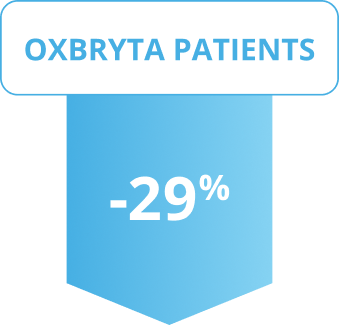

Results shown were at 24 weeks.
Patients on Oxbryta saw a significant decrease in average bilirubin levels compared to patients on placebo.
On average, reticulocyte levels decreased
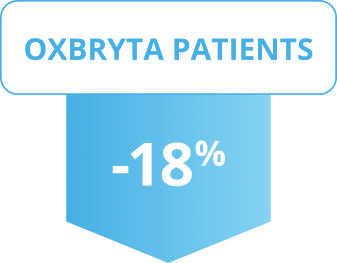
Results shown were at 24 weeks.
Patients on Oxbryta saw a significant decrease in their average reticulocyte counts, while patients on placebo saw their average counts increase.
"I like to ride my bike and go places, so I make sure to eat well, drink lots of water, and let my body rest every day."
– ABRAHAM
Actual Oxbryta patient
Actual Oxbryta patient
THE HOPE-KIDS 1 TRIAL STUDIED PATIENTS AGES 4 to less than 12 YEARS
Oxbryta 300 mg Tablets for Oral Suspension were studied in 45 patients ages 4 to less than 12 years old who received Oxbryta 300 mg Tablets for Oral Suspension (daily dose based on body weight) for 24 weeks.
The goal of this study
The HOPE-KIDS 1 clinical trial explored if Oxbryta helped increase hemoglobin by more than 1 gram per deciliter (g/dL) through 24 weeks of treatment.
- Most patients (80%) were taking hydroxyurea along with Oxbryta and continued to do so during the entire trial
OXBRYTA INCREASED HEMOGLOBIN
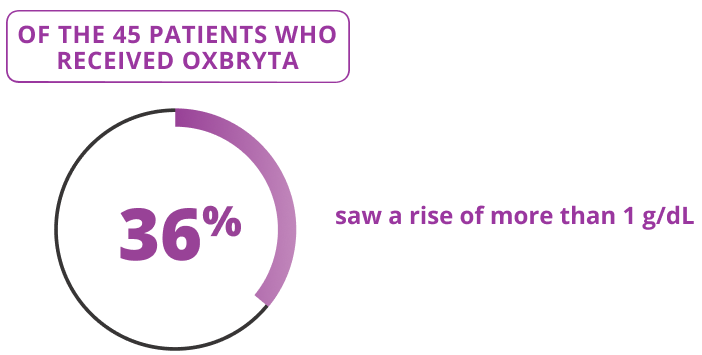
saw a rise of more than 1 g/dL
Talk to your healthcare provider to see if Oxbryta could be right for you.

Oxbryta Patient Brochure
Learn more about sickle cell and treatment with Oxbryta, and find specific questions you can ask your healthcare provider to get the conversation started.
DOWNLOAD"I like to draw and play on my tablet."
– HAJAR
Actual Oxbryta patient
Actual Oxbryta patient

